Breakdown of topics to come:
- Why We Run Keto Experiments
- How to Use Experiment Results
- What a Keto Experiment Looks Like
- What Can Sabotage or Bias Experiment Results?
- Step-by-Step Experiment How-to
- How to Get Involved
- Downloadable Experiment Checklist
- Summary: The Takeaways
Why We Run Keto Experiments
Often science hasn’t answered simple questions. Such as, can I eat this? Or, how will this product affect my ketones? That’s where we come in. Rather than guess the answers. We do experiments to equip you with keto knowledge.
It is also our hope that our experiments may one day inspire scientific research. So that our small-scale experiments are extended and verified by research establishments.
Whether it is to help you decide if something is keto. Or for how long the latest supplement will boost your ketones. Our main aim is to help you make informed decisions. Thus the primary outcomes of our experiments are:
- To inform people of our n = 1 discoveries
- To inspire and guide you to do your own Keto experiments
- To help answer your questions about Keto, fasting and health
- To establish hypothesises for larger, more thorough scientific investigations
What Our Keto Experiments Are NOT
Although we try to deliver accurate results. Our experiments are limited by sample size, equipment and budget. We cannot compete with research done by scientific establishments. We just don’t have the resources. But we can run small experiments in the hopes of helping you find answers. So our results should NOT be taken as:
- Medical advice
- True scientific research
- Scientific fact
- Universal to everyone
How to Use the Results
So now you know what our experiments are. You need to know how to use the results to personalise your ketosis. You can use our results to:
- Inform your eating, drinking and supplement purchases
- Decide on what to consume to maintain ketosis
- Run your own experiments – to check you get the same result.
What a Keto Experiment Looks Like
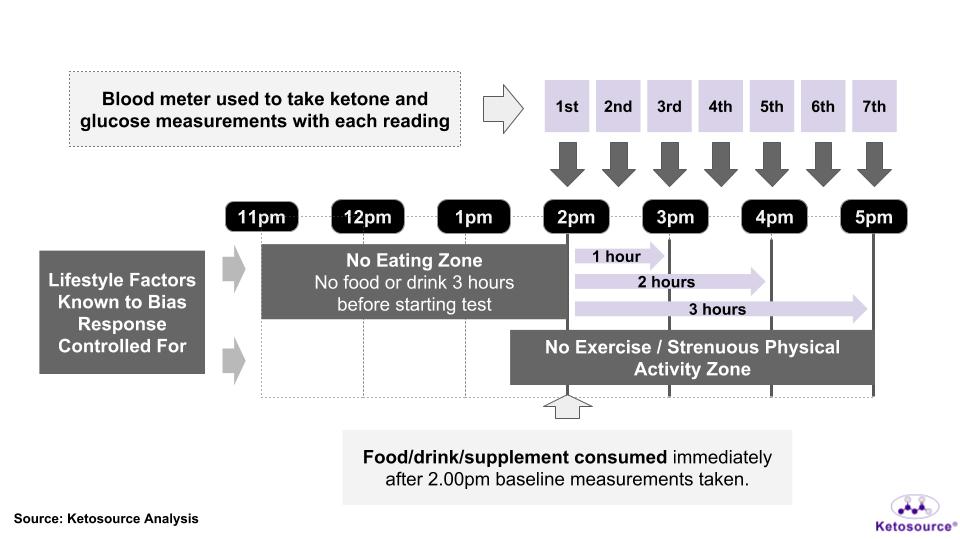
Here is an overview of a keto experiment. First lifestyle factors will be standardised which is up next. There will be a no eating zone for 3 hours before the start of the experiment. Along with a no exercise zone at least 30 minutes before and during the experiment.
First measurements will be taken before consuming anything. These will be the baseline readings for the day. Once the test substance is consumed the timer begins (NOTE: You will need 3 days of baselining before the real test). At each 30 minute intervals, you will measure glucose at least twice and ketones once.
The test will take 3 hours so plan to have some work on hand or for some entertainment in between readings. You have seen the big picture. Now we zoom into the details.
What Can Sabotage or Bias Experiment Results?
In all experiments, we have to try and standardise the results. This is in an effort to make the results reproducible and avoid confounders.
Reproducible = the experimental protocol must have enough detail to be copied by the reader to do the same experiment.
Confounders = something which can influence the results, other than what is being testing, the
All of the following factors are potential confounders. Each one can create a result by themselves on test day and therefore need to be standardised:
- Experiment start time
- Consumption time
- Sleep duration and quality
- Last meal
- Illness
- Weight
- Female menstrual cycle
- Exercise
- Fasting
- Equipment accuracy
So we try and control as many variables as we can. This means we have more certainty that only the item to be tested will be affecting our glucose and ketone measurements.
If you don’t control variables then who knows what caused the result. For instance, if you were to eat an apple an hour before your keto experiment. Do you think the new ketone bar you tested or the apple caused you ate beforehand caused a spike in your blood glucose?
Experiment Start Time
Blood glucose and ketones levels vary throughout the day. This is why you should try to do each experiment around the same time each day. 2 pm in the afternoon is a good time to run your experiment. Heres why:
It excludes hormonal influences on blood glucose in the morning. For instance, there is the well known early morning dawn phenomenon. This means certain people will see an abnormally high glucose reading. Due to a release of hormones (including cortisol, glucagon, epinephrine) which cause the liver to release glucose [note]Porcellati, Francesca, et al (2013). Thirty Years of Research on the Dawn Phenomenon: Lessons to Optimize Blood Glucose Control in Diabetes Diabetes Care 36.12: 3860-3862[/note].
This occurs mostly in diabetics, however, many factors can play a role in its appearance such as sleep deprivation. So non-diabetic individuals may also occasionally be subject to dawn phenomenon [note]Huang, Yuxin, et al. (2017). Poor Sleep Quality Is Associated with Dawn Phenomenon and Impaired Circadian Clock Gene Expression in Subjects with Type 2 Diabetes Mellitus International journal of endocrinology 36.12: 3860-3862.[/note]. By doing the experiment in the afternoon we remove this confounder.
An afternoon experiment also means insulin sensitivity should be in the moderate. Typically in a day, people are more insulin sensitive in the morning. While they are less insulin sensitive in the evening if not exercising.
Consumption Time
The time it takes to consume your food/drink/supplement should be based on how long it would normally take you to consume it. This time can make a difference on results since the faster you take something, the faster measurements will change and vice versa.
We generally consume our test food/drink/supplement within 10mins. This way you will clearly see if it causes an undesirable change in readings. If it usually takes you 30mins to drink a smoothie then make sure to incorporate this into your experimental protocol.
Sleep
Sleep can influence your results. It is well supported by the literature on how a lack of sleep causes an elevated glucose spike the next day. This is due to a lowered insulin sensitivity causing glucose to hang around in the blood. A lack of sleep will also cause the consequence of less satiety the next day. This may mean you eating more before the experiment, again affecting the results.
When tracking sleep there are two aspects to keep in mind: sleep quality and duration. This can be easily measured by asking the following questions before on the morning of each experiment and baselining day:
- Do you feel rested in the morning? – On a scale of 1-10.
- How long were you asleep for? – actually sleeping.
To track sleep duration either use a sleep tracking device such as a Fitbit or a sleep app on your phone. Alternatively, you can use a pen, paper and a timer. Note down when you go to bed and when you wake up. Or start a timer when you go to bed and stop it when you get up.
Record your sleep quality and duration. Before each test or baseline day (we will get into this later) check if you have had adequate sleep duration and quality.
If you sleep less than 7hrs you should not run the experiment the following day. If you do then be wary of the result. Likewise, if you woke in the morning feeling tired or a sleep tracker showed poor sleep quality.
Last meal
Not only what you ate, but when you ate your last meal will affect your results. It is best to eat a standard meal before each test. Fat, fibre and protein should make up the meal. Try to eat something which your system agrees with and can digest easily.
Inflammatory or hard to digest foods could cause irritation in your stomach or intestinal lining. An inflamed gut will not act the same as normal and can affect your glucose and ketone readings. So to help mitigate this you should either:
- Have a standard meal in the morning of the test day. Fast for at least 3 hours before the experiment
- Have a standard meal the night before the test day. Fast until consuming the test substance on the test day.
Illness
If you are ill this is not a good time to do the experiment. Your metabolism will be altered and blood glucose could be elevated as a result. Likewise, if you have stomach issues from a disturbed gut microbiota it would be better to fix this issue first.
There is no point in testing if you are unwell unless you plan on consuming the test item only in an unwell state.
Weight loss
If you are losing weight consistently than this may alter your results. Weight loss often boosts your ketones due to the breakdown and use of stored fat. This can lead to raised ketone and lower glucose readings at baseline before the experiment.
Weight loss could thus lead to a false negative result. For instance, a non-ketogenic food item may not kick you out of ketosis if you are losing weight. It is important to have a steady weight in the week of the experiment.
Female menstrual cycle
For female participants, time of the month can affect glucose and ketone readings. Thus if you are female and would like to do an experiment. Plan to do baselining and experiments before the end of your menstrual cycle.
Exercise
Exercise lowers your blood glucose and ketones. This is great in most circumstances however not before or while testing. So any form of physical activity other than typical day to day movement should be avoided as it may alter results.
Fasting
If you have been doing intermittent or prolonged fasting, you may see a greater rise in blood glucose when you eat your first meal. So if you break your fast with the consumable you are testing, you would need to repeat the same fast for each experiment. This is easy to do with intermittent fasting around 16hrs+. But prolonged fasting over a 24hrs may be hard to replicate.
In our experimental protocol, we advise you do not eat or drink anything with calories or artificial sweeteners at for 3hrs before the experiment starts time. This also holds true for natural sweeteners such as stevia due to the concern below.
Non-nutritive sweeteners
New research suggests that non-nutritive sweeteners such as stevia, aspartame and monk fruit extract can cause a greater blood glucose spike in response to your next meal [note]SL Tey, NB Salleh, J Henry and CG Forde (2016) Effects of aspartame-, monk fruit-, stevia- and sucrose-sweetened beverages on postprandial glucose, insulin and energy intake. International Journal of Obesity, volume 41, pages 450–457.[/note].
This means if you have a good dose one of these sweeteners earlier on in the day, your blood glucose may rise above what the testing item would usually cause.
How to Measure Glucose and Ketones
These are what gives you the results to interpret. All dual blood meters come with an instruction manual which should be followed carefully to obtain accurate results.
As a brief, clean and dry your hands thoroughly then rub the finger site to be used as a blood draw. You then insert the test strip into the device. When the meter automatically turns on, use the lancing device to draw blood from the outside of your finger.
Put the tip of the strip onto the blood and allow the blood to fill the line. Then wait for the result. Try to do multiple tests using the same site. So you will need to work quickly.
The blood meter works by basing glucose or βHB-ketone testing on the measurement of electrical current. This current comes from the reaction of glucose or βHB-ketone with the reagent of the strip. The meter measures the current, does a calculation, and spits out the blood glucose or β-ketone level on the screen.
Glucose – Always Double Check
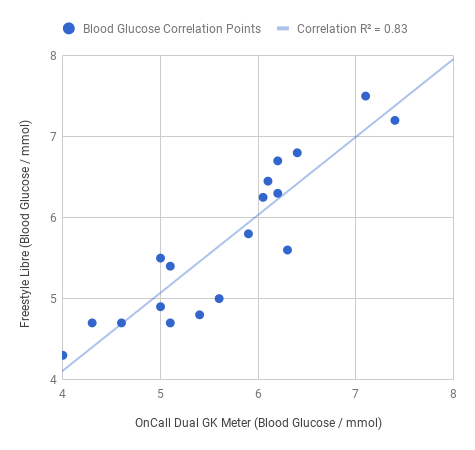
We need to double, and occasionally triple check, glucose readings. But why do we need to take multiple glucose readings at a time? To standardise our measurement results. The chart below provides evidence of how much a blood glucose readings vary from what they are meant.
What did we do to make this graph? In this series of experiments, we benchmarked the OnCall Dual glucose strips against the FreeStyle Optium blood glucose strips. We used the OnCall Dual blood meter and the Freestyle Libre blood meter/sensor. There were 24 glucose readings taken over the course of three experiments.
Each experiment took place over a separate day. And we used the average of two or three readings at a time. We measured the effect of consuming different fibres on blood glucose. The dots you see are where the values from both meters intersected.
The results gave us a correlation of 0.83 which is what the line on the graph represents. All you need to know is in a perfect world the values from each meter would line up along that line. So the closer they are the more accurate the results.
As an example when the Freestyle Libre blood sensor gave us a reading 5mmol/dL (90mg/dL) the OnCall Dual meter gave us a reading of 5.5mmol/L (99mg/dL). Then in a second experiment when again the Freestyle Libre blood sensor gave us a reading of 5mmol/dL (90mg/dL), the OnCall Dual meter gave us a reading of 4.9mmol/L (88.2mg/dL).
What Causes this Variation?
I know what you’re thinking. What causes this variation in readings? The very large range in which glucose meters can measure. For instance, the Abbott Precision Xtra can measure glucose readings between 2.0 and 26.8mmol/L (36 – 483mmol/L).
The FreeStyle Optium Neo can measure glucose readings between 1.1 and 27.8mmol/L (19.8 – 500mg/dL). Some meters have an even larger range of 0.6 to 33.3mmol/L (10.8 – 599.4 mg/dL).
Each Blood meter has to meet or exceed the minimum accuracy criteria set by ISO (International Organization for Standardization) [note]Freckmann, Guido, et al. (2015) Analytical performance requirements for systems for self-monitoring of blood glucose with focus on system accuracy: relevant differences among ISO 15197: 2003, ISO 15197: 2013, and current FDA recommendations. Journal of diabetes science and technology 9.4 885-894.[/note]:
- 95% of results >5.5mmol/L (99mg/dL) must fall within 15% of the reference method
- 95% of results <5.5mmol/L (99mg/dL) must fall within +0.83mmol/L (14.9 mg/dL) of the reference method
Therefore it is important to take at least two glucose readings using fresh glucose strips. Otherwise a reading that should be 5mmol/dL (90mg/dL) in a worst case scenario could come out as 5.83mmol/dL (104.9mg/dL) or 4.22mmol/dL (76.0mg/dL). Readings over 5.5mmol/dL can be even further from the true value.
Meter Accuracy
Glucose meters usually exceed the ISO accuracy standards. For instance, the Keto Mojo accuracy range is shown below:
| The accuracy range for readings under 75 mg/dL are: | The accuracy range for readings over 75 mg/dL are: |
| 100% within 15 mg/dL | 100% within 20% |
| 97% within 10 mg/dL | 98% within 15 % |
| 67% within 5 mg/dL | 96% within 10 mg/dL |
| 79% within 5 % |
There are additional factors that influence glucose readings. For instance, if readings are taken from different fingers. Since there will be fluid fluctuations between different finger regions.
Another factor is skin contaminants. That is why it is very important to wash your hands and have a clean skin area. Lastly, blood glucose changes rapidly. So taking readings just minutes apart will give you different results.
How Many Readings Should You Take?
So how do we determine if we need to do two or three readings? We do this with a simple rule. If two readings are within 5% or 0.2mmol/L (3.6mg/dL) of each other then you can take the average of the two readings. If however, the values have a difference over 5% or 0.2mmol/L (3.6mg/dL), then you will need to take a third reading.
Let us do an example. If your first reading was 4.8mmol/L (86mg/dL) then your next reading would need to be within 0.24mmol/L (4.3mg/dL). If you want to be really accurate your value should be no more than 5mmol/L (90mg/dL) and no less than 4.6mmol/L (82.8mg/dL) or you would need to do a third test.
So if the first reading was 5.5mmol/L (99mg/dL) and the second reading was 5.6mmol/L (100.8mg/dL) then you would calculate your average of the two values for this time point which is 5.55mmol/L (99.9mg/dL). If however, the second reading was 5.3mmol/L (95.4mg/dL) then we would need to do a third reading.
Let’s say the third value was 5.6mmol/L (100.8mg/dL). We would then take the average of all three values to give 5.46mmol/L (98.3mg/dL) for that time point. And voila that is how to measure blood glucose accurately.
Ketones
Ketones test strips have a much smaller range than glucose. Where glucose test strips can have a range of 24.8mmol/L. Ketone test strips only measure a range of around 8mmol.
Therefore, the variance is much lower due to the lower range. For instance, with the Keto Mojo ketone strips readings under 2 mmol/L will be within 0.3 mmol/L of laboratory results 90% of the time. Readings over 2mmol/L will be within 10% of the real value 81% of the time.
You can check your ketone strips in the documentation that comes with them in the box. In our testing, we have found that double checking is not necessary as results rarely differ at a single time point.
Step-by-Step Experiment How-to
What Equipment Do I Need?
To run your own keto experiments you will need some basic equipment. The setup will cost less than £100 and should last you through a number of experiments before you need to purchase more blood meter strips.
- Dual Glucose/ Ketone blood meter: Examples include ACON On Call GK Dual, Keto Mojo, Abbott Precision Xtra or Freestyle Optium Neo

- Other Tracking Equipment:
- Blood meter specific glucose AND ketone test strips – the meter will usually come with some but you will need extra
- Lancing device and lancet – to prick your finger
- Alcohol wipes or antibacterial soap
- Time Keeping and Recording devices
- Phone/stopwatch/pen and paper – to keep track of time and measurements.
- Sleep-tracking wearable or phone app would be useful but not essential to track the duration and quality of sleep.
Preparation
It is vital to establish what your glucose and ketones are at the time of day you will run the experiment. This means running a “fake” experiment for 3 days before the real experiment. We call this Baselining.
On baselining days you make sure to standardise as many lifestyle factors as possible. This means measuring sleep quantity and quality, eating the same pre-experiment meal or fasting for the same amount of time for 3 days.
On these days you would check your ketones and glucose as per the experimental protocol but without consuming anything. Just as you would if you were to run the real experiment with a test food/drink/supplement.
We do this to have baseline measurement for your ketones and glucose at each time point of the experiment. You then have your own ‘control’ results to compare your experiment results too.
If you do not know what your ketone and glucose readings are normally at say 3 pm. Then how do you know if what you tested caused a difference in your readings? This is why we do baselining.
How To Understand Your Baseline Measurements
Once completed you take the average of each of your readings over the course of the 3 days. For example, your 4 pm readings over the 3 baselining days are:
- Day 1: 5mmol/L, 5.2mmol/L = Average 5.1mmol/L
- Day 2: 4.4mmol/L, 4.7mmol/L, 4.5mmol/L = Average 4.53mmol/L
- Day 3: 4.4mmol/L, 4.5mmol/L = Average 4.45mmol/L
First, you calculate the average per reading so day 1 would be (5 + 5.2) / 2 = 5.1mmol/L. Day 2 = 4.53mmol/L. Day 3 = 4.45mmol/L. Then you add all the daily averages together and divide them by 3. So in this example, the average baseline glucose reading at 4 pm is 4.69mmol/L.
In summary, you follow the same experimental protocol and principles but do not consume anything at 2 pm for 3 days. You can then use your baseline results as a benchmark for any future experiments. Provided you don’t change diet and lifestyle too much.
What to test?
If you haven’t done already, you will need to decide on what food/drink/supplement is to be tested. Check out our keto experiments for examples:
Running the Experiment
For each time point, you take both ketone and glucose readings. You should use the same finger prick for both the ketone and glucose test. The measurements start with a baseline reading. This measurement is taken before consuming the test food/drink/supplement just before your experiment start time. In our example protocol, we would take a 1st baseline reading around 1:55 pm.
The measurement schedule looks like this:
- 1st reading: Baseline reading taken immediately before you start the test.
- Take the standard dose of agreed ‘testing item’
- 2nd reading: at 30 minutes
- 3rd reading: at 60 minutes
- 4th reading: at 90 minutes
- 5th reading: at 120 minutes
- 6th reading: at 150 minutes
- 7th reading: at 180 minutes
So if you take your test food/drink/supplement at 2 pm the next reading for blood glucose and ketones would be around 2:30 pm. The 3rd reading at 3 pm, the 4th at 3:30 pm etc. Up until 5 pm. Make sure to do no exercise between or before measurements.
Understanding the Results
Now you have your values, what do they mean? A good result would be a rise in ketones while glucose remains around the same level. You do not want to see your glucose go up and your ketones go down. This would indicate a non-ketogenic food/drink/supplement.
Glucose will normally rise a small amount to most foods. A glucose spike above 6mmol/L (108mg/dL) at any point in the experiment but especially at the 2-hour mark. Blood glucose should be kept under 120 mg/dl at all times – and as much as possible under 100 mg/dl.
You do not want your ketones to drop more than 0.25mmol/L. And you definitely need them to stay above 0.5mmol/L to stay in ketosis. Any reduction in ketones over 0.25mmol/L is considered a negative result for the test food/drink/supplement.
How to Get Involved
Do you have a burning question that needs answering? Do you want to be involved in our Keto Experiment? The answer is our Keto Experiment Community Portal.
Here you can submit experiment ideas. Vote on other people’s experiment ideas. Have your say in which experiments we do next. And learn from our previous experiment’s results.
Cut through the confusion, get data-driven results, get involved with the Keto Experiments Community.
Keto Experiment Checklist
To make things easy below is a downloadable protocol checklist. Print this off to make sure your keto experiments go on track. And remember these key points
- Baselining for 3 days before testing anything
- Take food/drink/supplement within 10mins
- Take measurements promptly
- Keep track of results
- Spreadsheet or paper
- Ask questions in the comments
- Get involved with the keto experiments community
Downloadable Keto Experiment Protocol Checklist
Summary: The Takeaways
- Keto experiments help you get personal results. We have unique biologies. Keto Experiments allow you to make more informed decisions on what actually works for you personally (as opposed to what the average population gets).
- Keto experiments are not science: Keto Experiments aren’t of the same quality or scale as research experiments done by scientific communities. They are helpful to understand your personal results, rather than conclusive.
- Standardise your experiment: Your results can be easily biased and give you false results if you aren’t careful. Standardise your experiment protocol following this guide to get the best results.
- Don’t forget to address the most common problems. Make sure to do your baseline testing and double/ triple check blood glucose readings.
- Interested in getting involved with Keto Experiments? Submit your own experiment ideas, upvote experiments you want us to do or browse the results from completed experiments in our Keto Experiments Community